Continuing Through-put Mo'orea Coral Extractions Five
10 DNA only extractions from the first set of Porites and Pocillopora Corals from Mo’orea, 7 to try again, and 3 of Kevin’s Astrangia samples
Using the Zymo Quick-DNA Miniprep Plus kit
Sample Prep
| Sample # | Type |
|---|---|
| 43 | Massive Porites |
| 48 | Pocillopora verrucosa |
| 54 | Massive Porites |
| 191 | Pocillopora verrucosa |
| 164 | Pocillopora verrucosa |
| 168 | Pocillopora verrucosa |
| 244 | Pocillopora verrucosa |
| 269 | Massive Porites |
| 275 | Massive Porites |
| 280 | Massive Porites |
| 210 | Massive Porites |
| 52 | Massive Porites |
| 272 | Massive Porites |
| 306 | Massive Porites |
| 285 | Massive Porites |
| 58 | Massive Porites |
| 39 | Massive Porites |
| H 15 | Astrangia poculata |
| H 21 | Astrangia poculata |
| H 27 | Astrangia poculata |
Mo’orea new samples
- Beads were poured into the new sample tubes. The new beads should be easier to pipette the liquid out of them as the do not get sucked up by the p20
- Samples were homogenized by vortexing for ~60 seconds for all samples
- Most of the liquid from the tubes was removed by pipetting. This was about 400µl. The tubes contained a small amount of liquid and un-homogenized tissue left, so 200µl of DNA/RNA shield was added to each tube and those were put back into the -20
- Following recommendations for samples in DNA/RNA Shield from the kit protocol, 200µl of Solid Tissue Buffer and 12µl of Proteinase K were added to each sample
- Samples were votexed, spun down, and incubated at 55 degrees C for 5 hours shaking at 600rpm
Mo’orea try again samples
- Whatever tissue was left was in ~300µl and the beads in the -20, another 100µl of DNA/RNA shield was added to make it roughly the same volume as the above samples
- Tubes were vortexed again for ~30 seconds to homogenize more
- 200µl of Blue Solid Tissue buffer and 12µl of Proteinase K were added to each sample (beads and all)
- Samples were votexed, spun down, and incubated at 55 degrees C for 5 hours shaking at 600rpm
- After digestion, as much liquid was removed as possible, all tubes still had some tissue in them. Sample 39 was very mucusy still
- Volume was about 500µl
Astrangia samples
- Samples were already lysed and stored as 500µl in the -80
- After the Mo’orea samples had digested for 5 hours, these samples were taken out to thaw
- 500µl of G-DNA binding buffer was added to each tube, vortexed, and spun down
- Extraction then followed the same way as the Mo’orea samples
Following DNA extraction went along exactly as previous post
Qubit
- Broad Range dsDNA Qubit protocol
- All samples were read twice
| Sample | DNA Standard 1 (RFU) | DNA Standard 2 (RFU) | DNA 1 (ng/µl) | DNA 2 (ng/µl) | Average DNA |
|---|---|---|---|---|---|
| 43 | 159 | 17132 | 46.8 | 47.4 | 47.1 |
| 48 | 159 | 17132 | 72.4 | 73.4 | 72.9 |
| 54 | 159 | 17132 | 106 | 109 | 107.5 |
| 191 | 159 | 17132 | 104 | 106 | 105 |
| 164 | 159 | 17132 | 72.8 | 75.2 | 74 |
| 168 | 159 | 17132 | 114 | 121 | 117.5 |
| 244 | 159 | 17132 | 87.4 | 88.8 | 88.1 |
| 269 | 159 | 17132 | 24.4 | 24.6 | 24.5 |
| 275 | 159 | 17132 | 24.8 | 23.4 | 24.1 |
| 280 | 159 | 17132 | 23.6 | 21.2 | 22.4 |
| 210 | 159 | 17132 | 10.8 | 10.9 | 10.8 |
| 52 | 159 | 17132 | 16.7 | 15.7 | 16.2 |
| 272 | 159 | 17132 | 38.8 | 38.6 | 38.7 |
| 306 | 159 | 17132 | 12.3 | 11.8 | 12 |
| 285 | 159 | 17132 | 8.76 | 9.56 | 9.16 |
| 58 | 159 | 17132 | 10.6 | 9.56 | 10.08 |
| 39 | 159 | 17132 | 26.4 | 25.8 | 26.1 |
| H 15 | 159 | 17132 | 51.4 | 51.6 | 51.5 |
| H 21 | 159 | 17132 | 13.1 | 12.8 | 13 |
| H 27 | 159 | 17132 | 40 | 40.4 | 40.2 |
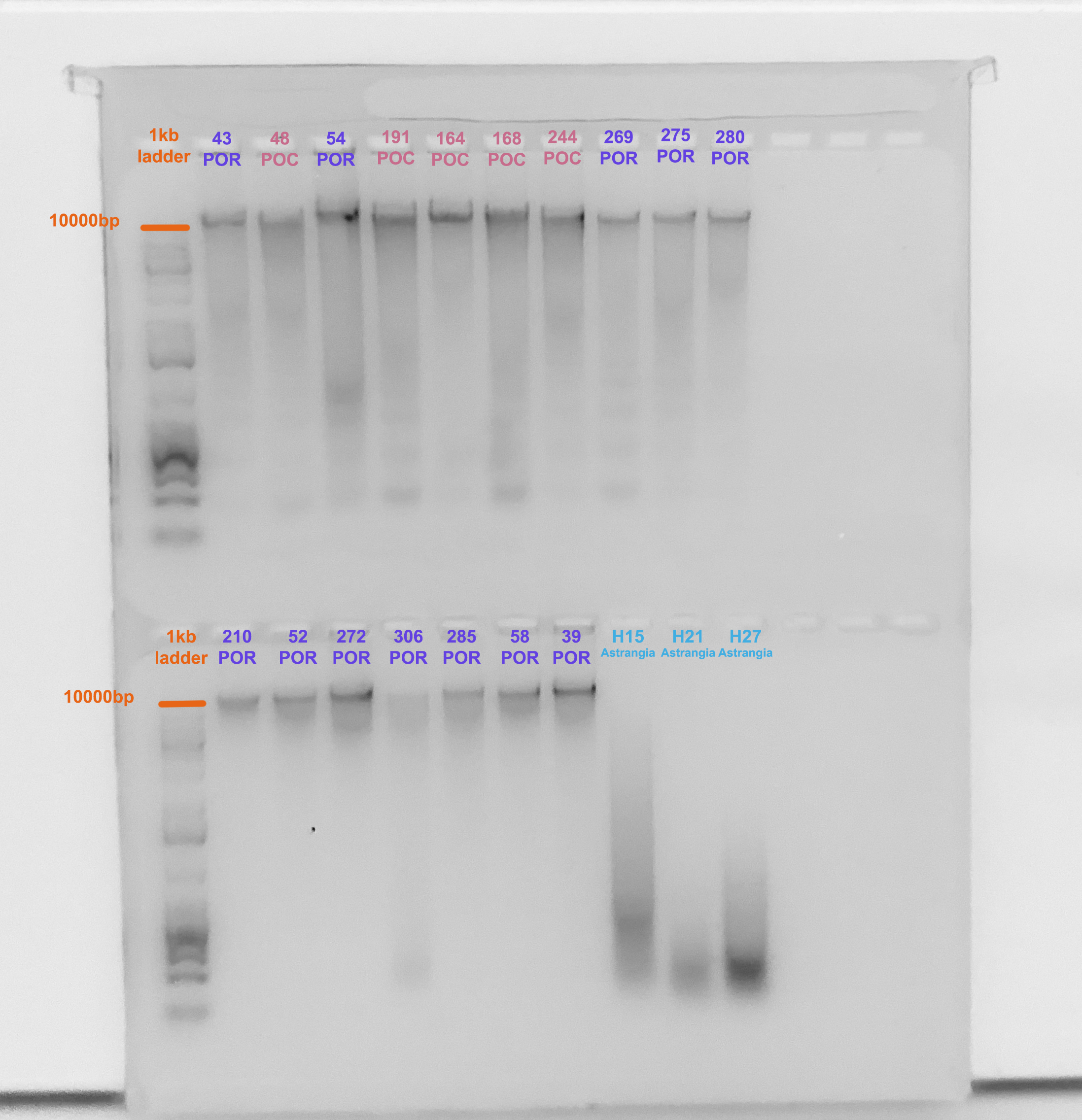
Gel Verification
- A 1.5% agarose gel was ran to check the integrity of the genomic DNA
- Following the PPP Lab protocol
Written on May 1, 2019